Canada authorises Pfizer COVID-19 vaccine for children aged 5-11
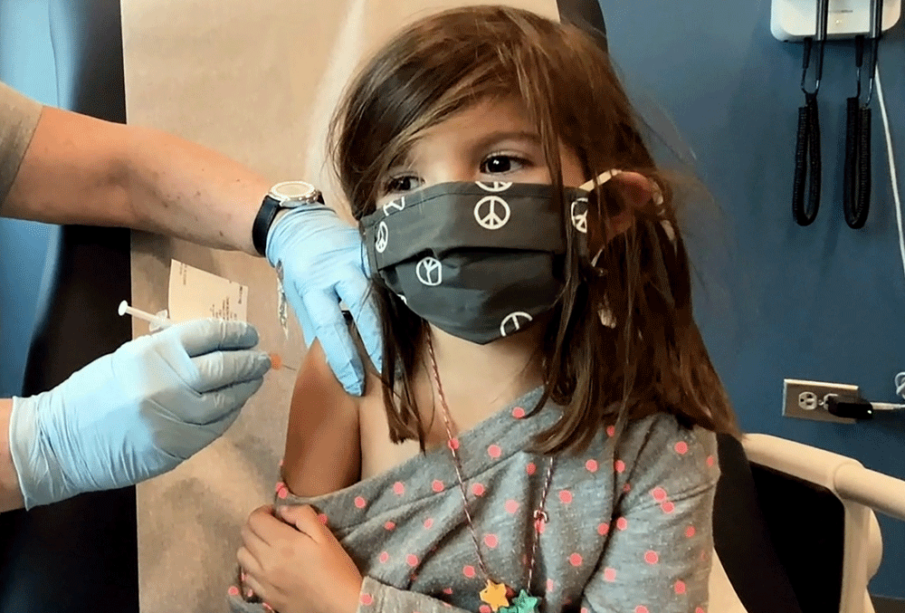 Bridgette Melo, 5, receives a reduced doses of the Pfizer BioNtech COVID-19 vaccine during a trial at Duke University in Durham, North Carolina, September 28, 2021. PHOTO BY SHAWN ROCCO/DUKE UNIVERSITY/ REUTERS
Bridgette Melo, 5, receives a reduced doses of the Pfizer BioNtech COVID-19 vaccine during a trial at Duke University in Durham, North Carolina, September 28, 2021. PHOTO BY SHAWN ROCCO/DUKE UNIVERSITY/ REUTERSCanada on Friday authorized the use of Pfizer Inc’s COVID-19 vaccine for children aged 5 to 11, paving the way for the inoculation of elementary school-age children across the country.
The shot is the first to be made available for young children in Canada. Officials had made clear for weeks that the decision would be favorable, noting that incidences of COVID-19 are highest in those under 12.
“After a thorough and independent scientific review of the evidence, the department has determined that the benefits of this vaccine for children between five and 11 years of age outweigh the risks,” Health Canada said in a statement.
Separately, Pfizer Canada said in a statement it would begin sending shipments imminently.
The vaccine will be given in two doses of 10 micrograms, three weeks apart. Adult doses contain 30 micrograms.
Prime Minister Justin Trudeau said last month that Ottawa had signed a deal with Pfizer to quickly receive 2.9 million doses of the vaccine once it was approved.
Health Canada said clinical trials showed the vaccine was 90.7% effective at preventing COVID-19 in those aged between 5 to 11 and no serious side effects were identified.
The U.S. Food and Drug Administration authorized use of the vaccine for those aged 5 to 11 on Oct. 29.
SOURCE: REUTERS